Research
The research is focused on developing chemical tools for drug discovery. My interest in this area emerged from the study of unnatural (mostly bicyclic) amino acids in the group of Prof. Igor Komarov back in 2000s. Currently, the focus is put on the design and synthesis of chemical building blocks (i.e., functionalized derivatives used to introduce a required structural fragment into the target compound) for early drug discovery. Other areas of interest include ultra-large chemically accessible compound libraries and cheminformatics tools to assess the compound’s lead-likeness. Recently, collaborations with material science people (metal-organic-frameworks, battery electrolytes) have emerged in my portfolio.
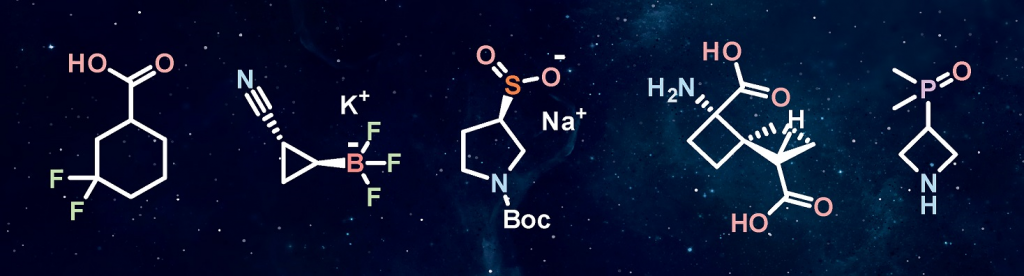
The design of building blocks is based on a combination of design concepts that appeared at the edge of organic and medicinal chemistry recently: target-oriented synthesis, diversity-oriented synthesis, and lead-oriented synthesis. The most significant of them, lead-oriented synthesis, focuses on the compound’s physicochemical and structural properties at the earliest steps of the study. That is why the aim is put at low-molecular-weight hydrophilic sp3-enriched carbo- and heterocyclic or heteroaromatic derivatives with one or two functional groups useful for parallel synthesis. As mentioned above, the research in this area started with the design and synthesis of bicyclic saturated amino acids and their analogs. To date, the range of chemotypes of interest is very large; some recent examples include:
- fluorinated sp3-enriched compounds;
- sp3-enriched boronic derivatives;
- saturated organosulfur building blocks;
- novel mono- and spirocyclic derivatives of small rings;
- motifs previously underrated in medicinal chemistry, e.g. phosphine oxides, sulfoximines, etc.
The main goal is extending the scope of chemical building blocks for early drug discovery by developing new chemotypes that follow (or possibly even create) the newest trends in medicinal chemistry, as well as by filling the “gaps” in the existing chemical space. Also, the effect of the developed building blocks or their major structural elements on the relevant physicochemical properties (such as pKa and LogP), as well as chemical reactivity in further parallel synthesis, are studied. This has been partially done already for the fluorinated sp3-enriched derivatives. In the field of chemically tractable ultra-large compound libraries, one of the current interests is the applications of machine learning methods to the chemical space created (in collaboration with specialists in the respective areas).
To date, I have co-authored over 270 papers, with an h-index of 31 (March 2026, Scopus).
